How does temperature affect oxygen solubility in water?
 Rachel Hickman
Rachel Hickman 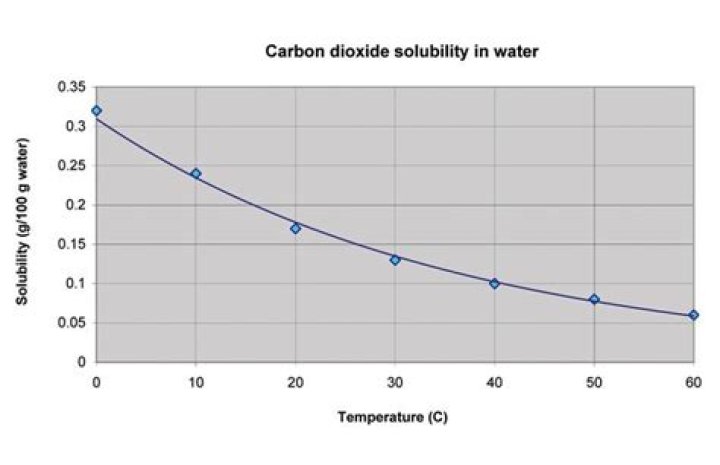
Also to know is, why does oxygen solubility in water decrease with temperature?
As the kinetic energy of the gaseous solute increases, its molecules have a greater tendency to escape the attraction of the solvent molecules and return to the gas phase. Therefore, the solubility of a gas decreases as the temperature increases.
Secondly, at what temperature is oxygen most soluble in water? 25oC
Keeping this in view, does temperature increase solubility of oxygen gas in water?
For many solids dissolved in liquid water, the solubility increases with temperature. The increase in kinetic energy that comes with higher temperatures allows the solvent molecules to more effectively break apart the solute molecules that are held together by intermolecular attractions.
Why does oxygen dissolve better in cold water?
Warm water holds less dissolved oxygen than cold water because the molecules are moving faster than in cold water and thereby allow oxygen to escape from the water. Therefore the warmer and saltier the water, the less dissolved oxygen it will contain.
Related Question Answers
How does temperature affect gas solubility?
As the temperature increases, the solubility of a gas decrease as shown by the downward trend in the graph . Increased temperature causes an increase in kinetic energy. The higher kinetic energy causes more motion in molecules which break intermolecular bonds and escape from solution.What are the 4 factors affecting solubility?
Factors affecting solubility- Temperature. Basically, solubility increases with temperature.
- Polarity. In most cases solutes dissolve in solvents that have a similar polarity.
- Pressure. Solid and liquid solutes.
- Molecular size.
- Stirring increases the speed of dissolving.
Why does dissolved oxygen decrease with temperature?
The actual amount of dissolved oxygen (in mg/L) will vary depending on temperature, pressure and salinity ¹. First, the solubility of oxygen decreases as temperature increases ¹. This means that warmer surface water requires less dissolved oxygen to reach 100% air saturation than does deeper, cooler water.Does solubility decrease with temperature?
The solubility of a solid in water increases with an increase in temperature. Gas solubility decreases as the temperature increases.Does solubility of oxygen increase with temperature?
Gases are usually more soluble at colder temperatures. For example, oxygen is more soluble in cold water than in hot water. The decrease in oxygen solubility with increased temperature [1] has serious consequences for aquatic life.Why is co2 less soluble in warm water?
Gases are generally more mobile than liquid molecules. At low temperatures they have less kinetic energy and thus are less able to escape. At high temperatures the liquid molecules are themselves moving fast and escaping, so the more mobile gas molecules get out even more quickly.What are the factors affecting solubility?
Factors affecting solubility- Temperature. Basically, solubility increases with temperature.
- Polarity. In most cases solutes dissolve in solvents that have a similar polarity.
- Pressure. Solid and liquid solutes.
- Molecular size.
- Stirring increases the speed of dissolving.
What are solubility curves used for?
Solubility curves is used to determine the mass of solute in grams per 100 grams of water(g/100gm) at the given temperature. Applications of curve; 1. It gives an idea about the ability of a solute to dissolve in water at different temperature.What is the effect of temperature and pressure on solubility?
The solubility of a solid may increase or decrease with increasing temperature, whereas the solubility of a gas decreases with an increase in temperature and a decrease in pressure.Are gases readily soluble in water at higher temperatures?
b) Carbon dioxide gas will be less soluble in water when the partial pressure is low. c) Gases are readily soluble in water at higher temperatures.Why must divers consider gas solubility?
Applications of Henry's Law of Solubility: Scuba divers must cope with high concentrations of dissolved gases while breathing air at high pressure underwater. Increased pressure increases the solubility of atmospheric gases in the blood. When the divers come towards the surface, the pressure gradually decreases.How can you increase the solubility of a drug?
One proven method of improving the solubility, and hence bioavailability, of your drug candidates is spray drying. The technique involves dissolving the drug in an organic solvent in the presence of a polymer. The resulting solution is then spray dried to form a dispersion (a spray-dried dispersion or SDD).Why is solubility important?
Solubility indicates the upper concentration a compound reaches in a solution. Solubility is a very important property in drug discovery and development, because concentration affects so many aspects of pharmacology (e.g., structure-activity relationships, efficacy, pharmacokinetics, toxicity).Will higher temperatures increase or decrease co2 solubility?
The solubility of the carbon dioxide in water increases as the temperature decreases. So the water in the bottle put in the freezer will have absorbed more CO2 from the air above it, and hence there will be less pressure of CO2. This is very important in the atmosphere system.What affects oxygen solubility in water?
The actual amount of dissolved oxygen (in mg/L) will vary depending on temperature, pressure and salinity ¹. First, the solubility of oxygen decreases as temperature increases ¹. Water at lower altitudes can hold more dissolved oxygen than water at higher altitudes.Does water absorb oxygen?
Oxygen is absorbed in water by direct diffusion and by surface-water agitation. Solubility of oxygen in water is so small and by diffusion process alone in still water, it was culculated that it would take 6 years for oxygen to diffuse from surface to a depth of 6 meters in quiet water.Is oxygen soluble or insoluble in water?
Oxygen is relatively insoluble in water, its solubility being only 264 μM at 25oC.What affects oxygen solubility?
Normally, oxygen solubility is strongly dependent on (1) the amount of dissolved electrolyte salt(s) (decreases at higher concentration of electrolyte), (2) temperature (decreases at higher temperatures), and (3) pressure (increases at higher pressure).What happens if oxygen is removed from water?
Water is one third oxygen; without it, the hydrogen turns into gaseous state and expands in volume. The oceans would evaporate and bleed into space. As oxygen disappears from the oceans' water, the hydrogen component becomes an unbound free gas.Does cold water dissolve more oxygen?
Dissolved oxygen, temperature, and aquatic life Cold water can hold more dissolved oxygen than warm water. In winter and early spring, when the water temperature is low, the dissolved oxygen concentration is high.Does warm water hold less oxygen?
The temperature and salinity of water influence how much oxygen it can hold. Warm water holds less dissolved oxygen than cold water because the molecules are moving faster than in cold water and thereby allow oxygen to escape from the water. (See Temperature section for more information.)Why does salt water hold less oxygen?
Salting Out. The ability of water to dissolve gases decreases with the addition of electrolytes. Salt ions attract water molecules leaving fewer hydrogen and oxygen ions available to capture and disassociate gas molecules.Why does gas dissolve better in cold water?
The more heat inside the water, the faster the water molecules will shoot out. A hot gas in a liquid will have a lot of energy and be able to escape the liquid, but a colder gas will have less energy to escape and be more likely to remain in the liquid making it more soluble.Why does oxygen dissolve in water?
Oxygen dissolves in surface water due to the aerating action of winds. Oxygen is also introduced into the water as a byproduct of aquatic plant photosynthesis. When dissolved oxygen becomes too low, fish and other aquatic organisms cannot survive. The colder water is, the more oxygen it can hold.Does pH affect dissolved oxygen levels in water?
Does pH affect a body of water's dissolved oxygen levels? We hypothesize that the dissolved oxygen levels decrease due to increasing levels of pH, thus inhibiting aquatic life that keeps dissolved oxygen levels high.Is there more oxygen in cold air?
Cooler air is more dense than warmer air. Warm air can actually hold more moisture because molecules are farther apart, making more room for moisture. Cold air is dense and compact; it's "thicker," so when you breathe in you get more oxygen. They get more oxygen and it's easier to cool their bodies.What are 3 factors that influence dissolved oxygen levels?
NATURAL FACTORS INFLUENCING DISSOLVED OXYGEN- Aquatic life- animals living in water use up dissolved oxygen.
- Elevation- the amount of oxygen in elevation increases.
- Salinity (saltiness)- Salty water holds less oxygen than fresh water.
- Temperature- cold water holds more dissolved oxygen than warm water.