What are wetting and non wetting liquids?
 Sebastian Wright
Sebastian Wright 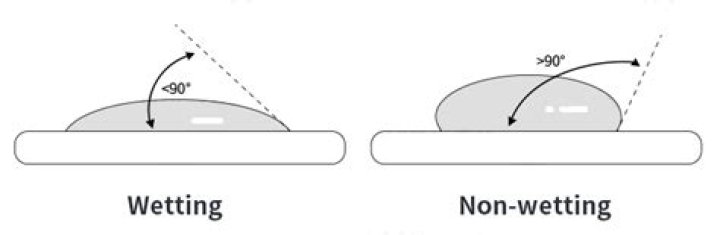
Also know, what is wetting liquid?
Wetting is the ability of liquids to form interfaces with solid surfaces. To determine the degree of wetting, the contact angle (q) that is formed between the liquid and the solid surface is measured. The smaller the contact angle and the smaller the surface tension, the greater the degree of wetting. Wetting.
Beside above, what liquids are not wet? According to information obtained from a scholarly database, Quora.com, “In a liquid-liquid interaction, such as water by itself, we can say that water is not wet, as molecules are all bound together and not wetting one another.”
Also, is water a wetting liquid?
Liquid water is not itself wet, but can make other solid materials wet. Wetness is the ability of a liquid to adhere to the surface of a solid, so when we say that something is wet, we mean that the liquid is sticking to the surface of a material.
Under what condition is a liquid said to wet a solid substance?
Hence if the contact angle between the liquid surface interface is zero, i.e., θ=00, then the degree of wetting is maximum and is said to be perfect wetting. In such case the solid-liquid interactions are much stronger than the liquid-liquid interactions. If 00<θ<900, then the degree of wettingis high.
Related Question Answers
Why is wetting important?
Wetting is important in the bonding or adherence of two materials. Wetting and the surface forces that control wetting are also responsible for other related effects, including capillary effects.What do wetting agents do?
Wetting agent, also called surfactant, chemical substance that increases the spreading and penetrating properties of a liquid by lowering its surface tension—that is, the tendency of its molecules to adhere to each other. See detergent; surfactant.What is wetting in soldering?
Solder wetting is the part of the process in which metal in the solder bonds with metal on the printed circuit board (PCB) or component. During the wetting process, the solder becomes fluid molten and can adhere properly to the component for an optimal solder joint.What is critical surface tension?
According to the Zisman method, the critical surface tension is the surface tension at which a liquid just completely wets a solid.What is wetting in dentistry?
236K subscribers. Wetting is the ability of a liquid to maintain contact with a solid surface, resulting from intermolecular interactions when the two are brought together. The degree of wetting (wettability) is determined by a force balance between adhesive and cohesive forces.How can we improve wettability?
An increase in adhesive force between the liquid and the solid or a decrease in the cohesive force (surface tension) within the liquid will result in greater wettability and a smaller contact angle.Why are wet surfaces more slippery?
wet surfaces are more slippery because the friction between our foot and the floor is reduced by the wet surface thus making it slippery. Wet surfaces are more slippery because the coating of liquid acts as a lubricant which reduces the frictional forces exerted between objects in contact.What is Wenzel state?
of a liquid droplet on a rough solid surface has long been associated with its. wetting state. When liquid drops are sitting on the top of the solid textures and. air is trapped underneath, they are in the Cassie state. When the drops impregnate the solid textures, they are in the Wenzel state.How do you test for wetting agent?
Another method is to place the liquid in a test tube and observe the liquid's meniscus (Figure 3). If the liquid contains a wetting agent, its adhesive forces are stronger than cohesive forces, which means the liquid molecules are more inclined to stick to the surface than other liquid molecules.Do wetting agents work?
Soil wetters, both liquid and granular; work by improving the absorption of water, reducing the surface tension of water and helping it to spread more evenly through the soil profile. Here at Garden Deva, we have found liquid wetting agents to be the most effective.How do you fix non wetting soil?
The best way to treat non wetting soils in a domestic lawn is with liquid wetting agents (not solid wetting agents) Liquid wetting agents do a couple of things – they help break down the waxy organic residues and they break the surface tension of the water, allowing it to penetrate into the soil.Can fire be wet?
Fire is a chemical reaction (rapid oxidation), not a substance. Water is a molecule (H2O). Since to be wet is to be covered in or saturated with water, water makes things wet. Water is wet, but fire is not dry.How can you prove water is wet?
“Water is wet because when something is wet, it has water on it and on a molecular level, water molecules are bonded on top of each other, therefore water is wet.” An extension to the previous argument that a couple of students brought up was that one water molecule alone is not wet, but when water molecules touch eachWhy is rain wet?
Frontal rainWarm air rises, so when it bumps into the cold air, it rises above it. So the warm air that has risen up in the atmosphere now begins to cool down. As a result, the water vapour (a gas) condenses into water (a liquid), forms a cloud, and eventually falls as raindrops.