We assume that the volume of the molecules themselves in a gas sample is negligible compared to the bulk volume of the gas sample: this helps us to explain why gases are so compressible. The Kelvin temperature is directly proportional to the average kinetic energy of the particles in a gas. Then, what does volume tell us about gas particles?
This law states that the volume and temperature of a gas have a direct relationship: As temperature increases, volume increases, when pressure is held constant. Heating a gas increases the kinetic energy of the particles, causing the gas to expand.
Likewise, what determines the volume of a gas? The volume of a gas is determined by the volume of the container it is in. Gases take the shape of their container and the volume of the container.
Beside this, what do scientists assume about gas particles size?
The main assumptions of the kinetic theory of gases are as follows: Gases are made up of particles (e.g. atoms or molecules). The size of these particles is very small compared to the distance between the particles. These particles are constantly moving because they have kinetic energy.
What is the volume of individual gas particles?
The volume occupied by individual molecules is negligible. - Gas molecules are constantly in random motion moving in straight paths, colliding with the walls of their container and with one another in perfectly elastic collisions.
Related Question Answers
What happens when gas is cooled?
If a gas is cooled, its particles will eventually stop moving about so fast and form a liquid. This is called condensation and occurs at the same temperature as boiling. Evaporation is dependent on individual particles gaining enough energy to escape the surface of the liquid and become gas particles. What are the 5 gases?
Examples of Gases - Hydrogen.
- Nitrogen.
- Oxygen.
- Carbon Dioxide.
- Carbon Monoxide.
- Water Vapour.
- Helium.
- Neon.
What are the 4 properties of gases?
Because most gases are difficult to observe directly, they are described through the use of four physical properties or macroscopic characteristics: pressure, volume, number of particles (chemists group them by moles) and temperature. What are the measurable properties of gas?
The measurable properties of gases are mass, volume, pressure and temperature. What happens to the particles in a gas when the gas is compressed?
What happens to the particles in a gas when the gas is compressed? The space between particles is reduced. Explain why heating a contained gas that is held at a constant volume increases its pressure. The increased kinetic energy of the particles causes collisions to occur more frequently and with more force. How do gas particles respond to an increase in volume?
Boyle's law states that the volume of a given mass of gas varies inversely with the pressure when the temperature is kept constant. An increase in pressure pushes the molecules closer together, reducing the volume. If the pressure is decreased, the gases are free to move about in a larger volume. Is Vapour a gas?
A vapor refers to a gas-phase material that that normally exists as a liquid or solid under a given set of conditions. As long as the temperature is below a certain point (the critical temperature; this varies for each substance), the vapor can be condensed into a liquid or solid with the application of pressure. Why do gas molecules move?
In gases the particles move rapidly in all directions, frequently colliding with each other and the side of the container. With an increase in temperature, the particles gain kinetic energy and move faster. Why are gases compressible but not liquid Class 9?
At room temperature and standard pressure, the average distance between two gas molecules is about 10 times the diameter of the complete molecule itself. Thus, gases get compressed easily as compared to solids and liquids when pressure is applied. Hence, gases are easily compressible but not liquids. What is different between ideal gas and real gas?
Ideal gases have mass and velocity. Real gas: Real gases are defined as the gases that do not obey gas laws at all standard pressure and temperature.
Write differences between real gas and ideal gas.
| Ideal gas | Real gas |
| It has high pressure | It has a pressure correction term in its equation and the actual pressure is less than ideal gas. |
Why are gas compressible but not liquid?
Why are gases compressible but not liquids? Answer: Gases are compressible because the intermolecular space is very large in gases, whereas liquids are not compressible because in liquids, the intermolecular space is less. Why can gasses be compressed?
The atoms and molecules in gases are much more spread out than in solids or liquids. They vibrate and move freely at high speeds. Gas can be compressed much more easily than a liquid or solid. What are the forces between particles in a gas?
Gas – In a gas, particles are in continual straight-line motion. The kinetic energy of the molecule is greater than the attractive force between them, thus they are much farther apart and move freely of each other. In most cases, there are essentially no attractive forces between particles. How gas is compressed into liquid in a gas barrel?
A gas can be converted into a liquid by increasing the pressure & decreasing temperature. So that the condensation occurs. You must also make the molecules closer so that it is a phase change from a gas to liquid. Why is liquid incompressible?
The amount of space (volume) the liquid occupies does not change (actually the volume does change but the change is very tiny). Liquids are always considered to be incompressible fluids, as density changes caused by pressure and temperature are small. Does a gas have a definite shape and volume?
A gas has neither a definite shape nor a definite volume. Like liquids, gases are fluids. The particles in a gas can move around one another freely. If a gas is released in a closed container, the gas particles will move in all directions and spread apart as they fill the container. What makes gas unique?
Gases have three characteristic properties: (1) they are easy to compress, (2) they expand to fill their containers, and (3) they occupy far more space than the liquids or solids from which they form. Does gas have mass and volume?
Gases have mass. The space between gas particles is empty. What is the relationship between volume and pressure?
It is summarized in the statement now known as Boyle's law: The volume of a given amount of gas held at constant temperature is inversely proportional to the pressure under which it is measured. What determines a liquids volume?
Subtract the weight of the container to get the weight of the liquid. Look up or calculate the density of the liquid, then determine the volume of the liquid by dividing the mass of the liquid by the density. Does gas have a fixed volume?
Gases don't have a fixed shape or a fixed volume. The particles move around all the time and spread out. This is why a gas fills its container. A gas can be compressed into a very small space – this pushes the particles closer together. What is volume in science?
Volume pertains to the three-dimensional space that is occupied by an object (i.e. solid, liquid, gas, or plasma). Volumes are determined by calculating the length, width, and height of the occupied space by an object. However, volume is different from mass. The mass is defined as the quantity of matter in an object. What happens to pressure when volume increases?
More collisions mean more force, so the pressure will increase. When the volume decreases, the pressure increases. This shows that the pressure of a gas is inversely proportional to its volume. What is the unit for volume of gas?
It has the SI unit of cubic metres per mole (m3/mol), although it is typically more practical to use the units cubic decimetres per mole (dm3/mol) for gasses, and cubic centimetres per mole (cm3/mol) for liquids and solids. What is the size of a gas particle?
In an ideal gas the particles have no size and no mutual attraction. They only have mass and motion -- which create pressure. What is the distance between particles in gas?
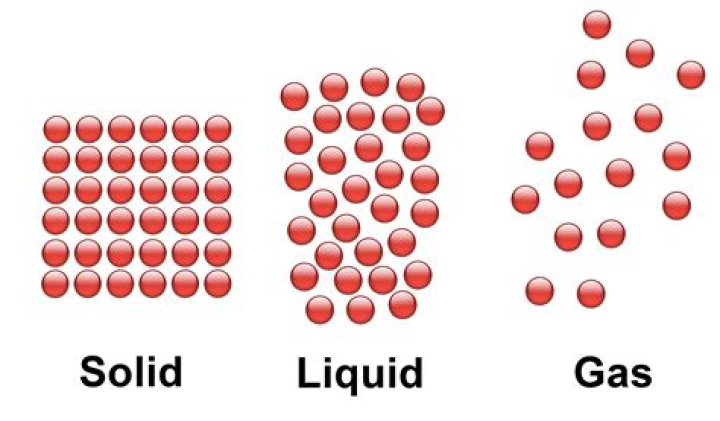
The particles in the solid are touching with very little space between them. The particles in a liquid usually are still touching but there are some spaces between them. The gas particles have big distances between them. How does the volume of gas particles compare to the volume of the space between gas particles?
Gases consist of very large numbers of tiny spherical particles that are far apart from one another compared to their size. In fact, the volume of the particles themselves is considered to be insignificant compared to the volume of the empty space. Gas particles are in constant rapid motion in random directions. What property of gas particles is measured by temperature?
The temperature of a gas is a measure of the average translational kinetic energy of the molecules. In a hot gas, the molecules move faster than in a cold gas; the mass remains the same, but the kinetic energy, and hence the temperature, is greater because of the increased velocity of the molecules. Which is an example of gas diffusion?
You can smell perfume because it diffuses into the air and makes its way into your nose. 2. Cigarette smoke diffuses into the air. In leaves, oxygen from the leaf cells diffuses out to the air. What is the ability of gas molecules to spread rapidly?
Gaseous particles are in constant random motion. Gaseous particles tend to undergo diffusion because they have kinetic energy. Diffusion is faster at higher temperatures because the gas molecules have greater kinetic energy. Effusion refers to the movement of gas particles through a small hole. What are gas laws in chemistry?
gas laws, laws that relate the pressure, volume, and temperature of a gas. These two laws can be combined to form the ideal gas law, a single generalization of the behaviour of gases known as an equation of state, PV = nRT, where n is the number of gram-moles of a gas and R is called the universal gas constant. Which graph shows the correct relationship between volume and pressure?
parabola
Which gas is more ideal at STP?
Answer: H2 is the more ideal gas.  Robert Young
Robert Young