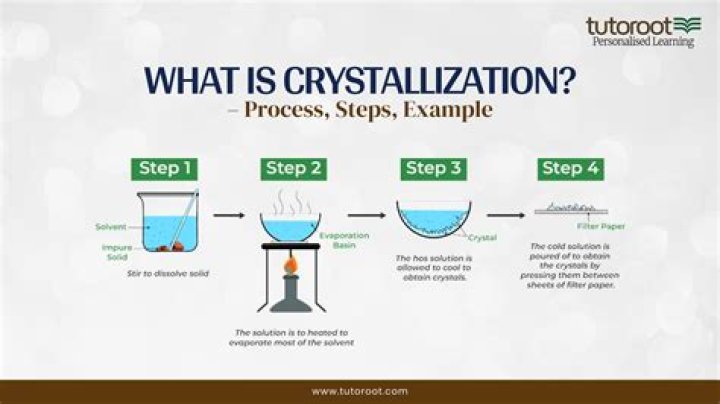
Any solid having definite geometrical shape is called crystal. The process of formation of a crystal is called crystallization. It is a technique in which a solute is separated from its saturated solution in the form of crystal. NaCl cannot purify by crystallization, because it is equally soluble in hot and cold water. Also question is, how is sodium chloride purified?
PURIFIED SALT (Sodium Chloride) Sodium chloride salt is produced directly from raw seawater through a state-of-the-art technology called Electro Dialysis, which is a membrane process. The material is then subjected to further evaporation/crystallization processes.
Subsequently, question is, why sodium chloride is not corrosive in nature? The Brainliest Answer! Sodium chloride forms when a sodium atom interacts with chlorine atoms and the sodium donates a negatively charged electron to the chlorine. This process makes sodium positively charged and chlorine negatively charged.
Also to know is, can NaCl purify water?
Disinfection of water is the most essential single step which can prevent epidemics of water borne diseases. In the present research work use of Sodium Chloride (NaCl) and Sodium Hypochlorite (NaOCl) is made for the disinfection of water. It is observed that for 30 min.
What happen when sodium chloride is heated?
When NaCl is heated, it gets hot then, at 801°C it melts and at 1413°C , it boils and when it is molten, It can conduct Electricity.
Related Question Answers
How do you make sodium chloride?
Fill a flask with 0.8 liters of water, weigh out the amount of sodium chloride you need, add it to the water and shake until it dissolves. To make a 1M solution, add 58.44 grams of salt; to make a 0.1M solution, add 5.84 grams; to make a 2M solution, add 116.88 grams and so on. What is the structure of sodium chloride?
NaCl
How is salt purified for human consumption?
The raw salt is refined to purify it and improve its storage and handling characteristics. This usually involves recrystallization during which a brine solution is treated with chemicals that precipitate most impurities (largely magnesium and calcium salts). How NaCl can be purified by common ion effect?
The common ion effect can make insoluble substances more insoluble. An example of the common ion effect is when sodium chloride (NaCl) is added to a solution of HCl and water. The hydrochloric acid and water are in equilibrium, with the products being H3O+ and Cl- . Then, some sodium chloride is added to the solution. What is sodium chloride used for?
Sodium chloride is the chemical name for salt. Sodium is an electrolyte that regulates the amount of water in your body. Sodium also plays a part in nerve impulses and muscle contractions. Sodium chloride is used to treat or prevent sodium loss caused by dehydration, excessive sweating, or other causes. How do I make purified salt?
Filtration - If the rock salt is one large chunk, grind it into a powder using a mortar and pestle or a coffee grinder.
- Add 30-50 milliliters of water to six heaping spatula scoops of rock salt.
- Stir to dissolve the salt.
- Place the filter paper in the mouth of the funnel.
How would you separate sodium chloride and potassium nitrate?
It can be done by sublimation. Heat the solution and allow it to cool and filter the precipitate of potassium chloride since it only dissolves in hot water. Heat the remaining filtrate until crystals of sodium chloride start forming and give it time to cool in order to form more crystals. Can I drink sodium chloride?
You should consult your doctor before adding more sodium chloride to your diet. Most people exceed the recommended amount, but people who drink excessive amounts of water, have persistent diarrhea, or participate in long endurance events may have sodium deficiency. In these cases, good oral hydration may help. Why is sodium chloride in water?
Salt (sodium chloride) in drinking water. Sodium is an essential mineral in our diet. It is commonly found in the form of sodium chloride (salt). Salt has no smell and it dissolves easily in water and gives water a “salty” taste at levels greater than 180 milligrams per litre. What salinity is safe to drink?
“The palatability of water with a total dissolved solids (TDS) level of less than about 600 mg/L is generally considered to be good; drinking-water becomes significantly and increasingly unpalatable at TDS levels greater than about 1000 mg/L.” Can drinking water reduce sodium level?
Drinking lots of water helps flush sodium from your kidneys; staying hydrated will also help you feel less bloated. Is low sodium water good for you?
Low sodium. Mineral water may be labeled “low in sodium“when its sodium content is below the limit of 20 mg/l. Reducing sodium intake is beneficial to maintaining normal blood-pressure. Gerolsteiner Naturell contains only 12 mg/l and is thus ideal for a low-sodium diet. Is salt water considered healthy to drink?
Seawater contains salt. While humans can safely ingest small amounts of salt, the salt content in seawater is much higher than what can be processed by the human body. Additionally, when we consume salt as part of our daily diets, we also drink liquids, which help to dilute the salt and keep it at a healthy level. Is chloride in water bad?
Chloride in drinking water is not harmful, and most concerns are related to the frequent association of high chloride levels with elevated sodium levels. What is the average salinity of tap water?
Fresh water - Less than 1,000 ppm. Slightly saline water - From 1,000 ppm to 3,000 ppm. Moderately saline water - From 3,000 ppm to 10,000 ppm. Is NaCl an acid or base?
NaCl is formed by the reaction of HCl and NaOH. Both are strong acids and bases. When a strong acid and a strong base react together the resultant is salt and water. Therefore NaCl is a salt. What is the pH of sodium chloride?
7
What type of bond is NaCl?
Salt or Sodium Chloride (NaCl) is a good example of a ionic bonding. Sodium (Na) has 1 valance electron and Chlorine (Cl) has 7 electrons in its outer orbit. If Sodium lost its valance electron, its next shell will be full. But that would also make Sodium a positive ion. What is the PKA of NaCl?
Sodium chloride does not have a pka. pka (or pkb) values only apply to substances that behave as weak acids and bases. Not salts, strong bases and strong acids. Is NaCl flammable?
(Dangerous when wet) Sodium is a FLAMMABLE SOLID which will ignite spontaneously in AIR or MOIST AIR and reacts violently with WATER or STEAM to produce flammable and explosive Hydrogen gas. Use dry chemicals appropriate for extinguishing metal fires such as graphite, soda ash or powdered sodium chloride. What elements are in sodium chloride?
Sodium's big claim to fame is that it's one of two elements in your table salt. When bonded to chlorine (Cl), the two elements make sodium chloride (NaCl). Sodium is also used as a salt in many fertilizers. What are the chemical properties of NaCl?
Formula and structure: The chemical formula of sodium chloride is NaCl and its molar mass is 58.44 g/mol. It is an ionic compound consisting of a sodium cation (Na+) and a chloride anion (Cl-). Solid NaCl has a crystalline structure, in which each Na+ ion is surrounded by six chloride ions in an octahedral geometry. Why is the formula for sodium chloride NaCl?
So then, sodium ions will attract chloride ions and form an ionic bond. By the way, chloride is the term used to designate the anion form of chlorine. The chemical formula for sodium chloride is NaCl, which means that for every sodium atom present, there is exactly one chloride atom. Does NaCl decompose when heated?
Some salts decompose readily upon heating such as bicarbonates salts. While some salts are stable when heated such as sodium chloride. However, if the cation in a salt has a high polarising power towards especially carbonates, hydroxide and nitrates ions, the salt will decompose to form a more stable salt. What happens when sodium chloride is added to water?
It can do this because water molecules carry an electric charge strong enough to disrupt the molecular bonds of the substances it dissolves. One such substance is sodium chloride (NaCl), or table salt. When you put salt in water, the sodium and chlorine ions separate and attach themselves to individual water molecules. What is the balanced equation for sodium chloride?
Chemical Equation Balancer Na + Cl2 = NaCl. What happens when Na and Cl combine?
The Sodium/Chlorine Reaction They combine as atoms, and separate as ions. When sodium and chlorine atoms come together to form sodium chloride (NaCl), they transfer an electron. The sodium (Na) atom transfers one electron to the chlorine (Cl) atom, so that they both have full outer shells. How sodium chloride is formed?
Sodium chloride is formed when sodium atoms interact with chlorine atoms. When this occurs, sodium will donate an electron (which is a negatively-charged particle) to chlorine. This makes sodium slightly positive and chlorine slightly negative. So then, sodium ions will attract chloride ions and form an ionic bond. How do you synthesize NaCl?
This type of reaction is represented by the general equation: A + B → AB. An example of a synthesis reaction is the combination of sodium (Na) and chlorine (Cl) to produce sodium chloride (NaCl). This reaction is represented by the chemical equation: 2Na + Cl2 → 2NaCl. Is sodium metal poisonous?
Reacts violently with water giving off flammable gas which may explode. At the time such a fire starts off, irritating and highly toxic gases are given off. After all, it is a flammable metal that is dangerous when untamed!  William Smith
William Smith